SAS is widely used in clinical research activities including:
- Managing and transforming data.
- Generating tabular and graphical summaries.
- Performing powerful statistical analyses such as safety and efficacy evaluations.
In addition, SAS provides a number of interfaces from which a user can select to work with the data. One of these newer interfaces, SAS® Studio, offers capabilities that can help busy users accomplish more in a day to ensure research studies are performed efficiently and that patients have access to the safest, most effective treatments possible.
SAS® Studio is a browser-based interface for writing and executing SAS programs. While visually similar to the familiar SAS windowing environment (aka “PC SAS”), the code can be executed by a SAS engine in the cloud, on your network or even on your PC with results returned to the browser.
SAS Studio ships with a variety of predefined “tasks” that provide point-and-click interfaces to perform common actions. Some of the tasks provided with SAS Studio include data tasks:
- Performing random sampling or standardizing data.
- Creating a variety of graph types.
- Using a variety of statistical analyses (including correlations and regressions).
More importantly, SAS Studio allows users to create their own tasks by easily designing an input form to support a set of code.
Why would you want to create a SAS Studio task?
A task provides a mechanism for exposing standardized code to a broader audience. A prime candidate for creating a task is to add a user interface to an existing macro. This eliminates the need to remember the parameters required for the macro – a user simply makes choices from the form that has been designed. This interface can include checks for interdependencies and enforce requirements for the user selections before the code is submitted to execute. This combination of features allows experienced users to reuse algorithms without having to refer to documentation or risk copy/paste issues from previous work and allows users with less programming experience to easily leverage validated code.
In the example below, we created a single SAS Studio task to make use of two popular published macros. The first macro performs a 1:N case-control propensity score matching and the second macro reports on the balance of the results from the first macro. In observational studies, it is common to match cases and controls based on similar propensity scores calculated using logistic regression of baseline covariates. This approach can reduce bias between treated and non-treated groups to more closely resemble a randomized clinical trial. A comparison of the distribution of baseline covariates between the resulting groups is needed to ensure the propensity score has been adequately specified.
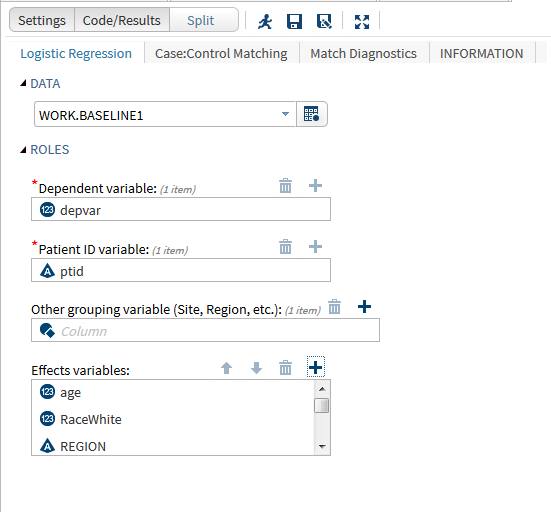
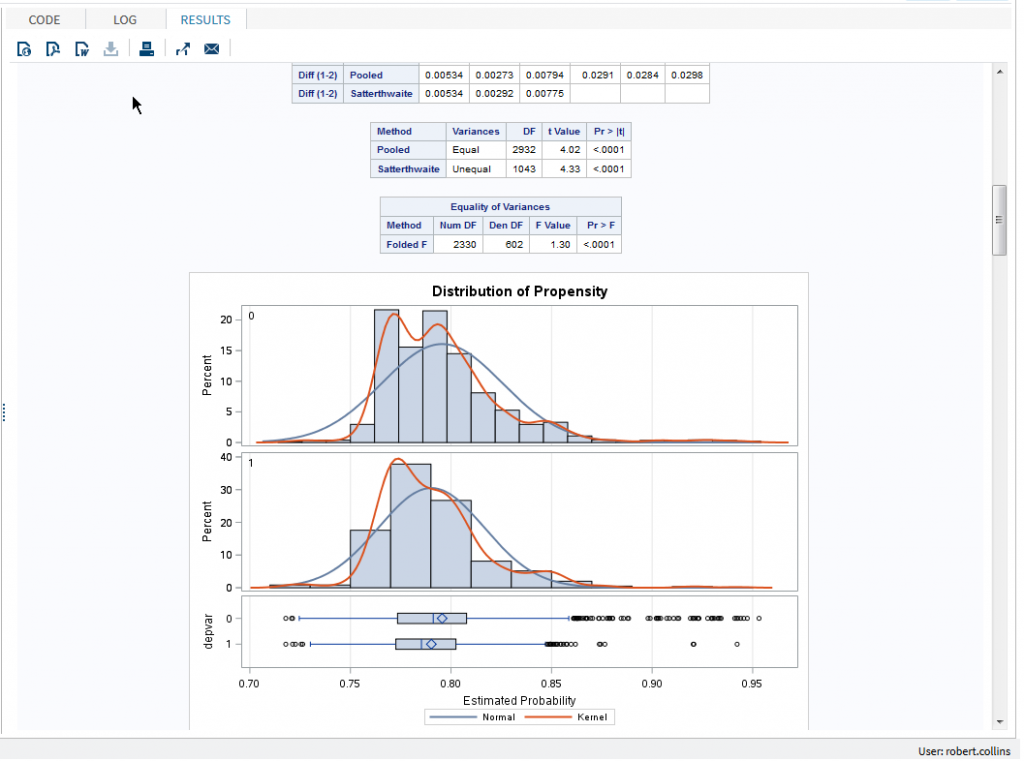
The same process can be applied to any other SAS code you may be using. At the upcoming DIA 2016 Annual Meeting in Philadelphia, we will present examples of SAS Studio Tasks for clinical data management, analytic data preparation, analytic methods and reporting including pushing results to other tools such as SAS Visual Analytics. Please stop by Booth 1825 to discuss how you may be able to use SAS Studio tasks in your organization.
For more details on SAS Studio and SAS Studio tasks, see the official documentation or refer to the links below:
- SAS Tech Report, May 2014.
- SAS Studio Tasks: Awesomeness You Might Be Missing from The Julia Group’s Annamaria’s Blog.
- Jedi SAS Tricks: DIY Tasks in SAS Studio from the SAS Learning Post blog.
For more details on propensity score matching, see the SAS Knowledge Base Usage Note 30971, Computing and matching observations based on propensity scores.
