For decades, researchers and programmers have used SAS to analyze, summarize, and report clinical trial data. Now Chris Holland and Jack Shostak have written the first comprehensive book on applying clinical research data and metadata to the Clinical Data Interchange Standards Consortium (CDISC) standards. What does this mean for you—the
Uncategorized
5 things you'll learn how to do with Implementing CDISC Using SAS
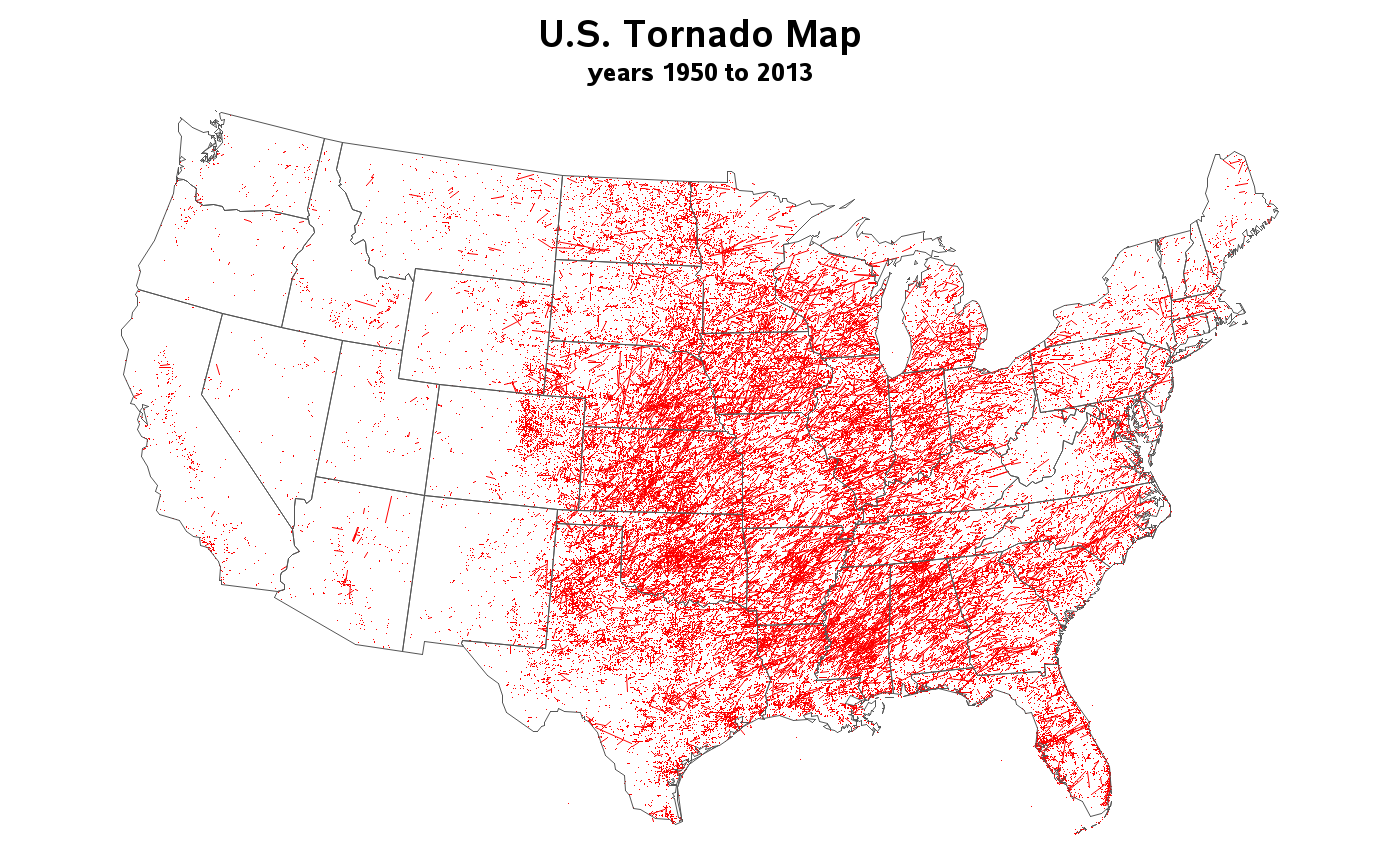
Tracking tornadoes with SAS
We had some fierce wind storms all across the United States yesterday, which even spawned some tornadoes! It looks like we weathered the storm OK here in Cary (but I still need to run out to the lake and make sure my pontoon/houseboat is still there, agh!). Anyway ... as with other
Tracking progress in your program with SAS Enterprise Guide: another trick
I've got a new trick that you can use to track progress in a long-running SAS program, while using SAS Enterprise Guide. I've previously written about the SYSECHO statement and how you can use it to update the Task Status window with custom messages. SYSECHO is a "global" statement in

